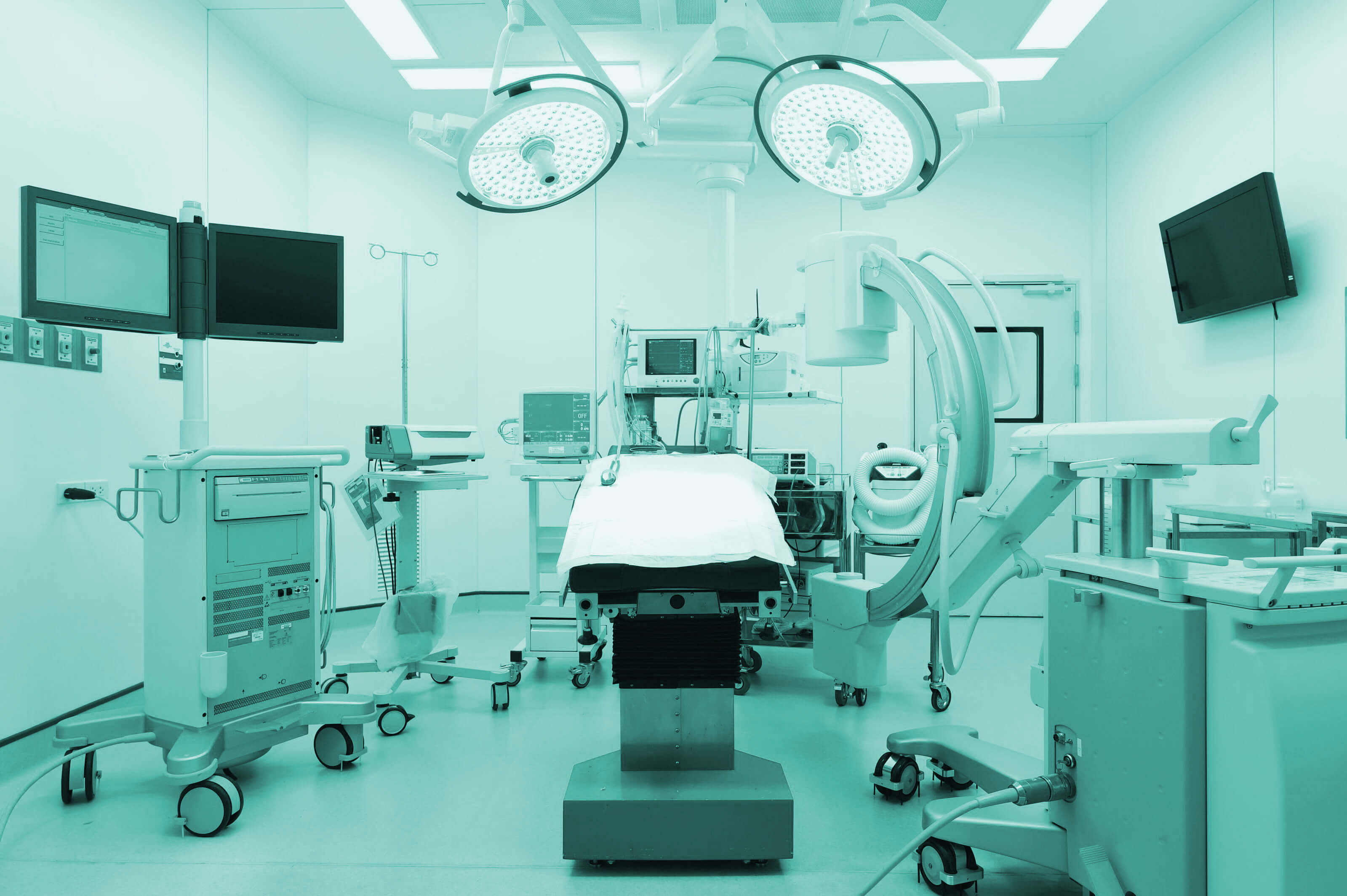
DQS offers various services in the field of approval of medical devices and certification of management systems in healthcare.
Choose your language
- South Africa English
- Global English
Local pages
- Bosnia and Herzegovina Bosanski
- Bulgaria български
- China 简体中文
- Hong Kong 中文
- Taiwan 繁體中文
- Croatia Hrvatski
- Czech čeština
- UAE English
- Australia English
- Aserbaidschan English
- Egypt English
- Ethiopia English
- United Kingdom English
- Hong Kong English
- India English
- Global English
- Lebanon English
- Malaysia English
- Nigeria English
- Pakistan English
- United States English
- Argentina Español
- Chile Español
- Colombia Español
- Spain Español
- Mexico Español
- El Salvador Español
- Iran ایرانی
- France Français
- Morocco Français
- Tunisia Français
- Germany Deutsch
- Hungary Magyar
- Indonesia Bahasa Indonesia
- Italy Italiano
- Japan 日本語
- Korea 한국어
- Poland Polski
- Brazil Português
- Portugal Português
- Russia Русский
- Serbia Srpski
- Slovakia Slovenský
- Turkey Türkçe
- Ukraine Українська
- Vietnam Tiếng Việt

Loading...
Any Questions?
We are here for you.